On the other hand, the ionic radius is half the distance between two ions that barely touch each other in a compound. Atomic radius is the distance between an atom’s nucleus and its outermost or valence electrons. For main group elements, the number of valence electrons usually ranges between 1 and 8 because eight electrons forms a complete octet. 
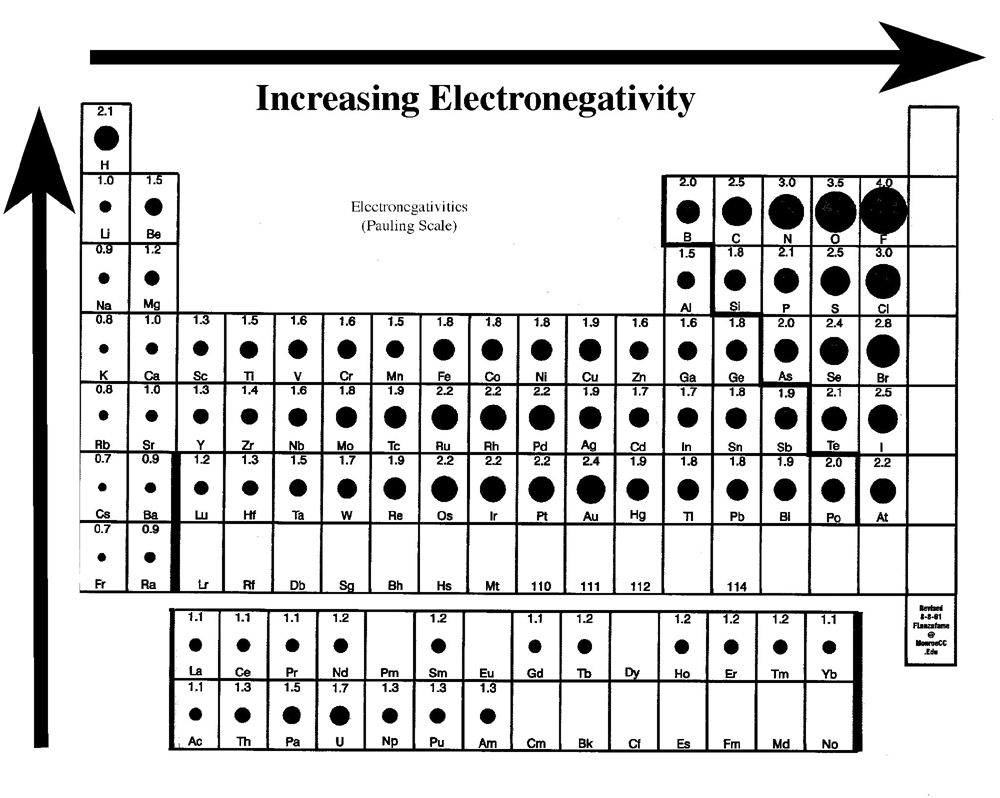
However, as you go down the periodic table, there are more filled shells, so the valence electron are further away from the nucleus, and so are not as tightly held in, increasing the atomic radius. Properties of Elements Based on Periodic Trends 1. As a 501(c)(3) nonprofit organization, we would love your help!Ĭreated by Ram Prakash. In the same group, the effective nuclear charge does not change. Khan Academy has been translated into dozens of languages, and 15 million people around the globe learn on Khan Academy every month. We provide teachers with tools and data so they can help their students develop the skills, habits, and mindsets for success in school and beyond. We offer quizzes, questions, instructional videos, and articles on a range of academic subjects, including math, biology, chemistry, physics, history, economics, finance, grammar, preschool learning, and more. Khan Academy is a nonprofit organization with the mission of providing a free, world-class education for anyone, anywhere. As we might predict, it becomes easier to add an electron across a series of atoms as the effective nuclear charge of the atoms increases. Values are not shown for groups 3 to 12 as well as period 7. Valency first increases and then decreases as we go from left to right in a period but remains the same in a group. The trend for electron affinity values across periods and down groups is shown in this version of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
